A) \[\begin{align} & C{{H}_{3}}-\underset{|}{\mathop{\overset{+}{\mathop{C}}\,}}\,-H \\ & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,C{{H}_{3}} \\ \end{align}\]
B) \[\begin{align} & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,C{{H}_{3}} \\ & C{{H}_{3}}-\underset{|}{\mathop{\overset{|}{\mathop{{{C}^{+}}}}\,}}\, \\ & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,C{{H}_{3}} \\ \end{align}\]
C) \[\overset{+}{\mathop{C}}\,{{H}_{3}}\]
D) \[C{{H}_{3}}\overset{+}{\mathop{C}}\,{{H}_{2}}\]
Correct Answer: B
Solution :
The most stable carbocation is t-alkyl carbocation because the order of stability of alkyl carbocation is t-alkyl> s-alkyl > p-alkyl > \[CH_{3}^{+}\] carbocation. This stability order is described with the help of hyperconjugation and inductive effect. On the Basis of hyper conjugation \[{{(C{{H}_{3}})}_{2}}\,\overset{+}{\mathop{C}}\,H\] shows six resonating structures due to presence of six \[\alpha \] C ? H bonds,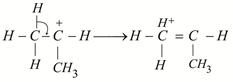 \[\underset{{}}{\longleftrightarrow}{{H}^{+}}\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,=\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,-H\underset{{}}{\longleftrightarrow}H-\underset{{{H}^{+}}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,=\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,-H\text{etc}\text{.}\] \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{{{C}^{+}}}}}\,\] shows nine resonating structures due to presence of nine \[\alpha \,C-H\] bonds. \[\overset{+}{\mathop{C}}\,\,{{H}_{3}}\] does not show the property of resonance while \[C{{H}_{3}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\] shows three resonating structures due to presence of three \[\alpha \,C-H\]bonds. Hence, larger number of resonating structures are possible in (2), so it is most stable. The above order of stability is also explained with the help of (+) \[l-\] effect of \[-C{{H}_{3}}\] group. More the number of \[-C{{H}_{3}}\] group more will be tendency to displace the electrons towards positive charged carbon of carbocation. Thus (+) charge is decreased or compensated i.e., positive charge is decreased and stability of carbocation is increased.
\[\underset{{}}{\longleftrightarrow}{{H}^{+}}\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,=\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,-H\underset{{}}{\longleftrightarrow}H-\underset{{{H}^{+}}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,=\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,-H\text{etc}\text{.}\] \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{{{C}^{+}}}}}\,\] shows nine resonating structures due to presence of nine \[\alpha \,C-H\] bonds. \[\overset{+}{\mathop{C}}\,\,{{H}_{3}}\] does not show the property of resonance while \[C{{H}_{3}}-\overset{+}{\mathop{C}}\,{{H}_{2}}\] shows three resonating structures due to presence of three \[\alpha \,C-H\]bonds. Hence, larger number of resonating structures are possible in (2), so it is most stable. The above order of stability is also explained with the help of (+) \[l-\] effect of \[-C{{H}_{3}}\] group. More the number of \[-C{{H}_{3}}\] group more will be tendency to displace the electrons towards positive charged carbon of carbocation. Thus (+) charge is decreased or compensated i.e., positive charge is decreased and stability of carbocation is increased.
You need to login to perform this action.
You will be redirected in
3 sec
