A) Acyl chloride > Acid anhydride > Ester > Amide
B) Ester > Acyl chloride > Amide >Acid anhydride
C) Acid anhydride > Amide, > Ester > Acyl chloride
D) Acyl chloride > Ester > Acid anhydride > Amide
Correct Answer: A
Solution :
Key Idea: The ease of nucleophilic substitution is depend upon the nature of leaving group. When the leaving tendency of a group in a compound is high, then the compound is more reactive towards nucleophilic substitution. The nucleophilic acyl substitution is completed in two steps as shown below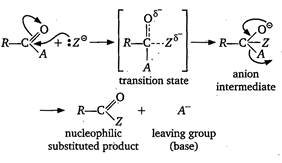 The reactivity of the compound may be explained on the basicity of the leaving group. A weaker base is a better leaving group. The basicity order is as: \[C{{l}^{-}}>RCO{{O}^{-}}>R{{O}^{-}}>NH_{2}^{-}\] Hence, the order of leaving tendency is \[C{{l}^{-}}>RCO{{O}^{-}}>R-{{O}^{-}}>NH_{2}^{-}\] and therefore, the order of reactivity of acyl compound is as:
The reactivity of the compound may be explained on the basicity of the leaving group. A weaker base is a better leaving group. The basicity order is as: \[C{{l}^{-}}>RCO{{O}^{-}}>R{{O}^{-}}>NH_{2}^{-}\] Hence, the order of leaving tendency is \[C{{l}^{-}}>RCO{{O}^{-}}>R-{{O}^{-}}>NH_{2}^{-}\] and therefore, the order of reactivity of acyl compound is as: You need to login to perform this action.
You will be redirected in
3 sec
