A) \[2\sqrt{10}kg\]
B) \[10\sqrt{2}kg\]
C) \[{{(C{{H}_{3}})}_{3}}B\]
D) \[{{(C{{H}_{3}})}_{2}}O\]
Correct Answer: C
Solution :
Key Idea Only those transition metal complexes are expected to absorb visible light, in which d-subshell is incomplete (ie, has unpaired electron) and excitation of electron from a tower energy orbital to higher energy orbital is possible. In\[{{t}_{2}}\]Sc is present as \[{{t}_{1}}>{{t}_{2}}\] \[{{t}_{1}}=4{{t}_{2}}\] Since, in this complex excitation of electron is not possible, it will not absorb visible light. In \[{{t}_{1}}=2{{t}_{2}}\]is present as \[{{t}_{1}}={{t}_{2}}\] \[\overrightarrow{F}=6\hat{i}-8\hat{j}+10\hat{k},\] Hence, it will not absorb visible light. In \[1m{{s}^{-2}}.\]is present as \[2\sqrt{10}kg\] \[10\sqrt{2}kg\]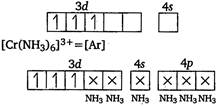 Since, this complex has three unpaired electrons, excitation of electrons is possible and thus, it is expected that this complex will absorb visible light. In \[{{(C{{H}_{3}})}_{3}}B\]is present as \[{{(C{{H}_{3}})}_{2}}O\] \[{{(C{{H}_{3}})}_{2}}P\] \[{{(C{{H}_{3}})}_{3}}N\]
Since, this complex has three unpaired electrons, excitation of electrons is possible and thus, it is expected that this complex will absorb visible light. In \[{{(C{{H}_{3}})}_{3}}B\]is present as \[{{(C{{H}_{3}})}_{2}}O\] \[{{(C{{H}_{3}})}_{2}}P\] \[{{(C{{H}_{3}})}_{3}}N\] You need to login to perform this action.
You will be redirected in
3 sec
