A) \[{{\text{ }\!\![\!\!\text{ Ni(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{\text{2+}}}\]
B) \[{{\text{ }\!\![\!\!\text{ Zn(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{\text{2+}}}\]
C) \[{{\text{ }\!\![\!\!\text{ Cr(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{\text{3+}}}\]
D) \[{{\text{ }\!\![\!\!\text{ Co(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{6}}}\text{ }\!\!]\!\!\text{ }}^{\text{3+}}}\]
Correct Answer: A
Solution :
Outer orbital complex utilizes \[nd\] orbitals for bonding and exhibit paramagnetic behaviour, only if there present unpaired electrons. In \[{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}:\] \[N{{i}^{2+}}=[Ar]3{{d}^{8}}4{{s}^{o}}\] So, this is an outer orbital complex having paramagnetic character. In\[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}:\]\[Z{{n}^{2+}}=[Ar]\,3{{d}^{10}}\]
So, this is an outer orbital complex having paramagnetic character. In\[{{[Zn{{(N{{H}_{3}})}_{6}}]}^{2+}}:\]\[Z{{n}^{2+}}=[Ar]\,3{{d}^{10}}\] 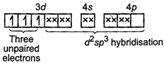 Because of the involvement of (n - 1) d, i. e., 3d orbital-in hybridization, it is an inner orbital complex. Its nature is paramagnetic because of the presence of three unpaired electrons. In \[{{[CO{{(N{{H}_{3}})}_{6}}]}^{3+}}:\]\[C{{o}^{3+}}=[Ar]\,3{{d}^{6}}\]
Because of the involvement of (n - 1) d, i. e., 3d orbital-in hybridization, it is an inner orbital complex. Its nature is paramagnetic because of the presence of three unpaired electrons. In \[{{[CO{{(N{{H}_{3}})}_{6}}]}^{3+}}:\]\[C{{o}^{3+}}=[Ar]\,3{{d}^{6}}\] 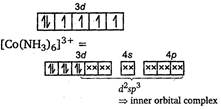 As all the electrons are paired, it is a diamagnetic complex.
As all the electrons are paired, it is a diamagnetic complex.
You need to login to perform this action.
You will be redirected in
3 sec
