A) \[{{K}_{B}}=2{{K}_{C}}\]
B) \[{{K}_{D}}=10\,mg\]
C) \[{{K}_{B}}+{{K}_{C}}={{K}_{D}}\]
D) \[({{K}_{B}}+{{K}_{C}})<{{K}_{D}}\]
Correct Answer: C
Solution :
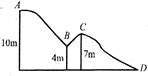
: Potential energy (P.E) at \[A=wgh=10mg\] P.E at \[B=mg\times 4=4mg\] \[\therefore \] K.E at \[B={{K}_{B}}=(10-4)mg=6mg\] P.E at \[C=mg\times 7=7mg\] \[\therefore \]K.E. at \[C={{K}_{C}}=(10-7)mg=3mg\] P.E at \[D=mg\times 0=zero\] K.E. at\[D={{K}_{D}}=(10-0)mg=10\,mg\] Option [a] \[{{K}_{B}}=6\,mg\] \[{{K}_{C}}=3\,mg\] \[\therefore \]\[{{K}_{B}}=2{{K}_{C}}\]is correct. Option [b] \[{{K}_{D}}=10\,mg\] is correct. Option [c] \[{{K}_{B}}=6\,mg\] \[{{K}_{C}}=3\,mg\] \[{{K}_{D}}=10\,mg\] \[{{K}_{B}}+{{K}_{C}}=(6+3)=9\,mg\ne {{K}_{D}}\] Hence this option is incorrect. Option [d] \[{{K}_{B}}+{{K}_{C}}=6+3=9\,mg,{{K}_{D}}=10\,mg\] \[\therefore \] \[{{K}_{B}}+{{K}_{C}}<{{K}_{D}}\].This is correct.You need to login to perform this action.
You will be redirected in
3 sec
