A) square planar
B) trigonal bipyramidal
C) tetrahedral three-dimensional
D) octahedral three-dimensional
Correct Answer: B
Solution :
: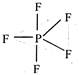 Trigonal bipyramidal geometry of\[P{{F}_{5}}\]molecule resulting from\[s{{p}^{3}}d\]hybridisation of valence shell orbitals of central P atom.
Trigonal bipyramidal geometry of\[P{{F}_{5}}\]molecule resulting from\[s{{p}^{3}}d\]hybridisation of valence shell orbitals of central P atom.
You need to login to perform this action.
You will be redirected in
3 sec
