A) \[{{C}_{6}}{{H}_{5}}C{{H}_{2}}CHO\]
B) \[{{C}_{6}}{{H}_{5}}CHO\]
C)
![]()
D)
![]()
Correct Answer: A
Solution :
Key Idea Benzoin condensation reaction is given by aromatic aldehydes in which \[-CHO\]group is directly linked to the benzene nucleus. In \[{{C}_{6}}{{H}_{5}}C{{H}_{2}}CHO\] the \[-CHO\] group is not attached directly to the benzene nucleus. So, it does not undergo benzoin condensation. While in , and the \[-CHO\] group is directly linked to benzene nucleus. So, these compound undergo benzoin condensation. Note- In benzoin condensation two molecules of aromatic aldehyde, on heating in the presence of ethanolic KCN get condensed to form benzoin.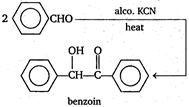
You need to login to perform this action.
You will be redirected in
3 sec
