A)
B)
![]()
C) \[C{{H}_{3}}-C{{H}_{2}}-CHO\]
D)
![]()
Correct Answer: B
Solution :
\[\underset{propionic\text{ }acid}{\mathop{C{{H}_{3}}C{{H}_{2}}COOH}}\,\xrightarrow{SOC{{l}_{2}}}\underset{\begin{smallmatrix} \left( A \right) \\ propanoyl\text{ }chloride \end{smallmatrix}}{\mathop{C{{H}_{3}}C{{H}_{2}}COCl}}\,\] \[\xrightarrow[\begin{smallmatrix} Rosenmund \\ reaction \end{smallmatrix}]{Pd/BaS{{O}_{4}}}\underset{\begin{smallmatrix} (B) \\ propanal \end{smallmatrix}}{\mathop{C{{H}_{3}}C{{H}_{2}}.CHO}}\,\]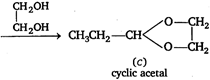 When propionic acid reacts with thionyl chloride \[(SOC{{l}_{2}})\], then \[-OH\] group of carboxylic acid replaced by \[-Cl\] group and propanoyl chloride is formed which reacts with \[Pd/BaS{{O}_{4}}\] and compound , ie, propanal is obtained (Rosenmund reaction). This compound , ie, propanal reacts with ethylene glycol, gives a cyclic acetal compound .
When propionic acid reacts with thionyl chloride \[(SOC{{l}_{2}})\], then \[-OH\] group of carboxylic acid replaced by \[-Cl\] group and propanoyl chloride is formed which reacts with \[Pd/BaS{{O}_{4}}\] and compound , ie, propanal is obtained (Rosenmund reaction). This compound , ie, propanal reacts with ethylene glycol, gives a cyclic acetal compound .
You need to login to perform this action.
You will be redirected in
3 sec
