A) \[C{{H}_{3}}-C{{H}_{2}}-C\equiv CH\]
B) \[C{{H}_{3}}-C\equiv CH\]
C) \[C{{H}_{3}}-C\equiv C-C{{H}_{3}}\]
D) \[CH\equiv CH\]
Correct Answer: B
Solution :
Key Idea According to Markownikoffs rule ?The addition of a reagent to an unsymmetric (alkene or alkyne) takes place in such a way that the negative part of the reagent will be attached to the carbon atom which contain lesser number of hydrogen atom. \[A+HOCl\xrightarrow{{}}C{{H}_{3}}-\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-CHC{{l}_{2}}\] \[C{{H}_{3}}C{{H}_{2}}-C\equiv CH+\overset{-}{\mathop{H}}\,O\overset{+}{\mathop{Cl}}\,\xrightarrow{{}}\] \[C{{H}_{3}}C{{H}_{2}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C}}\,=CHCl\xrightarrow{HOCl}\]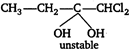 \[\xrightarrow{-{{H}_{2}}O}C{{H}_{3}}C{{H}_{2}}\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-CHC{{l}_{2}}\] \[C{{H}_{3}}C\equiv CH+\overset{-}{\mathop{H}}\,O\overset{+}{\mathop{Cl}}\,\xrightarrow{{}}\] \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C}}\,=CHCl\]
\[\xrightarrow{-{{H}_{2}}O}C{{H}_{3}}C{{H}_{2}}\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-CHC{{l}_{2}}\] \[C{{H}_{3}}C\equiv CH+\overset{-}{\mathop{H}}\,O\overset{+}{\mathop{Cl}}\,\xrightarrow{{}}\] \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C}}\,=CHCl\] 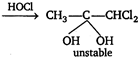 \[\xrightarrow{-{{H}_{2}}O}C{{H}_{3}}-\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-CHC{{l}_{2}}\] \[C{{H}_{3}}-C\equiv C\,.\,\,C{{H}_{3}}+\overset{-}{\mathop{H}}\,O\overset{+}{\mathop{Cl}}\,\xrightarrow{{}}\] \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C}}\,=\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{3}}\xrightarrow{HOCl}\]
\[\xrightarrow{-{{H}_{2}}O}C{{H}_{3}}-\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-CHC{{l}_{2}}\] \[C{{H}_{3}}-C\equiv C\,.\,\,C{{H}_{3}}+\overset{-}{\mathop{H}}\,O\overset{+}{\mathop{Cl}}\,\xrightarrow{{}}\] \[C{{H}_{3}}-\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C}}\,=\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{3}}\xrightarrow{HOCl}\] 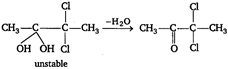 \[CH\equiv CH+HOCl\xrightarrow{{}}\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,H=CH\,.\,OH\]
\[CH\equiv CH+HOCl\xrightarrow{{}}\underset{\begin{smallmatrix} | \\ Cl \end{smallmatrix}}{\mathop{C}}\,H=CH\,.\,OH\] 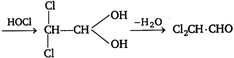 Therefore, answer is correct, ie, A is\[C{{H}_{3}}C\equiv CH\].
Therefore, answer is correct, ie, A is\[C{{H}_{3}}C\equiv CH\].
You need to login to perform this action.
You will be redirected in
3 sec
