A) butylene
B) butadiene
C) isobutylene
D) propylene
Correct Answer: B
Solution :
Key Idea Isolated dienes : They contain two double bonds separated by two or more saturated carbon atoms. Cumulative dienes : They contain two double bonds at adjacent positions. Conjugated dienes : They contain alternating system of single and double bonds. Conjugated diene is butadiene because butadiene contain alternating system of single and double bonds. \[C{{H}_{2}}=CH-CH=C{{H}_{2}}\] Therefore, conjugated double bond is present in butadiene. While in butylene, isobutylene and propylene only one double bond is present. So, these are not dienes and do not contain conjugated double bond. \[C{{H}_{3}}C{{H}_{2}}CH=C{{H}_{2}}\] butylene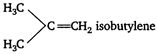 \[C{{H}_{3}}CH=C{{H}_{2}}\] propylene
\[C{{H}_{3}}CH=C{{H}_{2}}\] propylene
You need to login to perform this action.
You will be redirected in
3 sec
