A) monobasic acid and dibasic acid
B) monobasic and tribasic acid
C) monobasic acid and a salt
D) dibasic acid and tribasic acid
Correct Answer: A
Solution :
\[P{{I}_{3}}\] on hydrolysis gives, phosphorus acid and hydroiodic acid. \[P{{I}_{3}}+3{{H}_{2}}O\xrightarrow{{}}\underset{dibasic}{\mathop{{{H}_{3}}P{{O}_{3}}}}\,+\,\,\,\underset{monobasic}{\mathop{3HI}}\,\] \[{{H}_{3}}P{{O}_{3}}\] is a dibasic acid. The structure of \[{{H}_{3}}P{{O}_{3}}\] is as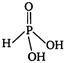 In \[{{H}_{3}}P{{O}_{3}}\] the two hydrogen atom are ionisable and one hydrogen atom is non-ionisable. Hence, it is a dibasic acid. While in HI one hydrogen atom is ionisable. So, it is a monobasic acid.
In \[{{H}_{3}}P{{O}_{3}}\] the two hydrogen atom are ionisable and one hydrogen atom is non-ionisable. Hence, it is a dibasic acid. While in HI one hydrogen atom is ionisable. So, it is a monobasic acid.
You need to login to perform this action.
You will be redirected in
3 sec
