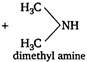
A) \[{{(C{{H}_{3}})}_{2}}NH+C{{H}_{3}}COOH\]
B) \[{{(C{{H}_{3}})}_{2}}NCOOH+C{{H}_{4}}\]
C) \[2C{{H}_{3}}OH+C{{H}_{3}}CON{{H}_{2}}\]
D) \[2C{{H}_{3}}N{{H}_{2}}+C{{H}_{3}}COOH\]
Correct Answer: A
Solution :
Key Idea Amides on acidic hydrolysis give acid and an amine (or ammonia) \[RCON{{H}_{2}}+HOH\xrightarrow{{{H}^{+}}}RCOOH+N{{H}_{3}}\] \[RCONH\,.\,C{{H}_{3}}+HOH\xrightarrow{{{H}^{+}}}\]\[RCOOH+C{{H}_{3}}N{{H}_{2}}\] The refluxing of \[{{(C{{H}_{3}})}_{2}}N.\,\,COC{{H}_{3}}\] with acid gives acetic acid and dimethyl amine.
You need to login to perform this action.
You will be redirected in
3 sec
