A) Rubber
B) Nylon-6
C) Nylon-6, 6
D) Nylon-6, 10
Correct Answer: A
Solution :
Rubber is a natural ploymer. It is a polymer of isoprene. \[\underset{isopropene}{\mathop{C{{H}_{2}}-\overset{\begin{smallmatrix} C{{H}_{3}} \\ | \end{smallmatrix}}{\mathop{C}}\,-CH=C{{H}_{2}}}}\,\] \[\underset{natural\text{ }rubber}{\mathop{\xrightarrow{{}}\,\,\left( -C{{H}_{2}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,=CH-C{{H}_{2}}- \right)}}\,\] Nylon-6, Nylon-6 6 and Nylon-6, 10 are synthetic polymer or man made polymers. Nylon-6 It is the polymer of caprolactum.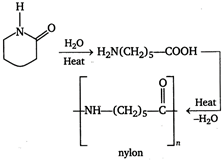 Nylon-6, 6 It is the polymer of hexamethylenediamine and adipic acid. \[\underset{\begin{smallmatrix} hexamethylene \\ diamine \end{smallmatrix}}{\mathop{n{{H}_{2}}N-{{(C{{H}_{2}})}_{6}}N{{H}_{2}}}}\,+nOH-\]\[\underset{adipic\text{ }acid}{\mathop{\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{2}}{{)}_{4}}-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-OH}}\,\] \[\underset{nylon,\text{ }6,\text{ }6}{\mathop{\xrightarrow{{}}\,\,\,{{\left[ -\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{N}}\,-{{(C{{H}_{2}})}_{6}}-\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{N}}\,-\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-{{(C{{H}_{2}})}_{4}}-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,- \right]}_{n}}}}\,\] Nylon-6, 10 It is polymer of hexamethylenediaminie and sebacic acid. \[n{{H}_{2}}N-{{(C{{H}_{2}})}_{6}}N{{H}_{2}}+nHOOC+nHOOC-\]\[{{(C{{H}_{2}})}_{8}}-COOH\] \[\xrightarrow[-2n{{H}_{2}}O]{}\] \[\underset{nylon,\text{ }6,\text{ }10}{\mathop{\xrightarrow{{}}\,\,\,{{\left[ -NH-{{(C{{H}_{2}})}_{6}}-\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{N}}\,-\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-{{(C{{H}_{2}})}_{8}}-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,- \right]}_{n}}}}\,\]
Nylon-6, 6 It is the polymer of hexamethylenediamine and adipic acid. \[\underset{\begin{smallmatrix} hexamethylene \\ diamine \end{smallmatrix}}{\mathop{n{{H}_{2}}N-{{(C{{H}_{2}})}_{6}}N{{H}_{2}}}}\,+nOH-\]\[\underset{adipic\text{ }acid}{\mathop{\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{2}}{{)}_{4}}-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-OH}}\,\] \[\underset{nylon,\text{ }6,\text{ }6}{\mathop{\xrightarrow{{}}\,\,\,{{\left[ -\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{N}}\,-{{(C{{H}_{2}})}_{6}}-\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{N}}\,-\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-{{(C{{H}_{2}})}_{4}}-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,- \right]}_{n}}}}\,\] Nylon-6, 10 It is polymer of hexamethylenediaminie and sebacic acid. \[n{{H}_{2}}N-{{(C{{H}_{2}})}_{6}}N{{H}_{2}}+nHOOC+nHOOC-\]\[{{(C{{H}_{2}})}_{8}}-COOH\] \[\xrightarrow[-2n{{H}_{2}}O]{}\] \[\underset{nylon,\text{ }6,\text{ }10}{\mathop{\xrightarrow{{}}\,\,\,{{\left[ -NH-{{(C{{H}_{2}})}_{6}}-\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{N}}\,-\underset{\begin{smallmatrix} || \\ O \end{smallmatrix}}{\mathop{C}}\,-{{(C{{H}_{2}})}_{8}}-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,- \right]}_{n}}}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
