A) Perkins reaction
B) Wurtz-Fittig reaction
C) Wurtz reaction
D) Williamsons synthesis
Correct Answer: D
Solution :
Williamsons synthesis This reaction is used to prepare both symmetrical and unsymmetrical ethers by treating alkyl halide with sodium alkoxide or sodium phenoxide. \[\underset{\begin{smallmatrix} ethyl \\ chloride \end{smallmatrix}}{\mathop{{{C}_{2}}{{H}_{5}}Cl}}\,+\underset{\begin{smallmatrix} sodium \\ ethoxide \end{smallmatrix}}{\mathop{{{C}_{2}}{{H}_{5}}ONa}}\,\xrightarrow{{}}\underset{diethyl\text{ }ether}{\mathop{{{C}_{2}}{{H}_{5}}O{{C}_{2}}{{H}_{5}}}}\,+NaCl\]Perkins reaction Benzaldehyde on heating with acetic anhydride in the presence of sodium acetate and the hydrolysis of product obtained gives \[\alpha ,\beta \] unsaturated acid, cinnamic acid.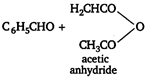
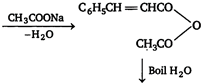 \[\underset{cinnamic\text{ }add}{\mathop{{{C}_{6}}{{H}_{5}}CH=CHCOOH}}\,+C{{H}_{3}}COOH\] Wurtz reaction Alkyl halide react with sodium in the presence of dry ether to form alkanes. \[C{{H}_{3}}C{{H}_{2}}Cl+2Na+ClC{{H}_{2}}C{{H}_{3}}\] \[\xrightarrow{Dry\text{ }ether}\underset{butane}{\mathop{C{{H}_{3}}C{{H}_{2}}\,.\,\,C{{H}_{2}}C{{H}_{3}}}}\,+2NaCl\] Wurtz-Fittig reaction Haloalkanes react with haloarenes in the presence of sodium and anhydrous ether to form alkyl benzene.
\[\underset{cinnamic\text{ }add}{\mathop{{{C}_{6}}{{H}_{5}}CH=CHCOOH}}\,+C{{H}_{3}}COOH\] Wurtz reaction Alkyl halide react with sodium in the presence of dry ether to form alkanes. \[C{{H}_{3}}C{{H}_{2}}Cl+2Na+ClC{{H}_{2}}C{{H}_{3}}\] \[\xrightarrow{Dry\text{ }ether}\underset{butane}{\mathop{C{{H}_{3}}C{{H}_{2}}\,.\,\,C{{H}_{2}}C{{H}_{3}}}}\,+2NaCl\] Wurtz-Fittig reaction Haloalkanes react with haloarenes in the presence of sodium and anhydrous ether to form alkyl benzene. 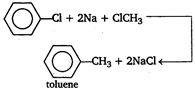
You need to login to perform this action.
You will be redirected in
3 sec
