A) m-bromophenol
B) o-and p-bromophenol
C) p-bromophenol
D) 2, 4, 6-tribromophenol
Correct Answer: B
Solution :
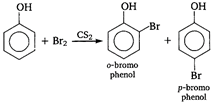 In presence of non-polar solvent \[(C{{S}_{2}})\] the ionisation of phenol is suppressed. The ring is slightly activated and hence, mono substitution occurs. On the other hand with Br^ water phenol gives 2, 4, 6-tribromo phenol.
In presence of non-polar solvent \[(C{{S}_{2}})\] the ionisation of phenol is suppressed. The ring is slightly activated and hence, mono substitution occurs. On the other hand with Br^ water phenol gives 2, 4, 6-tribromo phenol. 
You need to login to perform this action.
You will be redirected in
3 sec
