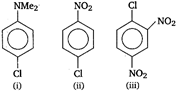
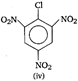
 is
is
A) i > ii > iii > iv
B) ii > i > iii > iv
C) iv > iii > ii > I
D) iii > iv > ii > i
Correct Answer: C
Solution :
Nucleophile attacks readily to the compound that has least electron density. Electron withdrawing groups like ?Cl, \[N{{O}_{2}}\] etc decrease the electron density while electron releasing groups like NMe2 increase the electron density at benzene nucleus. Compound (iv) due to the presence of four electron withdrawing groups has the least electron density and thus, is more reactive towards nucleophilic substitution. Hence, the correct order of reactivity towards nucleophilic substitution is as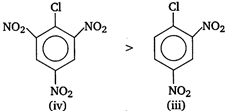
You need to login to perform this action.
You will be redirected in
3 sec
