A) \[{{C}_{6}}{{H}_{5}}COC{{H}_{2}}COOH\]
B) \[{{C}_{6}}{{H}_{5}}CH\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{C}}\,OOH\]
C) \[{{C}_{6}}{{H}_{5}}\underset{\begin{smallmatrix} | \\ N{{H}_{2}} \end{smallmatrix}}{\mathop{C}}\,HCOOH\]
D) \[{{C}_{6}}{{H}_{5}}COCOOH\]
Correct Answer: A
Solution :
If keto group is at p-position then decarboxylation takes place simply by heating, p -keto acid is very reactive in decarboxylation reaction because it produces a highly stable carbanion.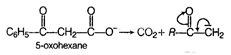
You need to login to perform this action.
You will be redirected in
3 sec
