A) 1
B) 2
C) 3
D) 0
Correct Answer: D
Solution :
In \[Ni{{(CO)}_{4}}\], there is no unpaired electron. It is diamagnetic. Oxidation state of Ni in \[[Ni{{(CO)}_{4}}]\] complex compound is zero. \[Ni=3{{d}^{8}}4{{s}^{2}}\]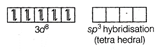 \[[Ni{{(CO)}_{4}}]\]
\[[Ni{{(CO)}_{4}}]\] 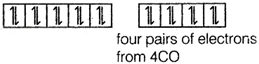
You need to login to perform this action.
You will be redirected in
3 sec
