A) Pyramidal, \[s{{p}^{3}}\]
B) Octahedral, \[s{{p}^{3}}{{d}^{2}}\]
C) Tetrahedral, \[s{{p}^{3}}\]
D) None of these
Correct Answer: C
Solution :
The shape of \[BH_{4}^{-}\] is tetrahedral because in\[BH_{4}^{-}\], boron is \[s{{p}^{3}}\] hybridised.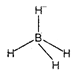
You need to login to perform this action.
You will be redirected in
3 sec
