A) 2-methylbutene
B) but-1-ene
C) but-2-ene
D) 1,3-butadiene
Correct Answer: A
Solution :
\[\underset{\underset{A}{\mathop{2-methyl\text{ }butene}}\,}{\mathop{C{{H}_{3}}-C{{H}_{2}}-\underset{\begin{smallmatrix} | \\ C{{H}_{3}} \end{smallmatrix}}{\mathop{C}}\,=C{{H}_{2}}+{{O}_{3}}\xrightarrow{{}}}}\,\]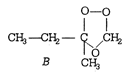
You need to login to perform this action.
You will be redirected in
3 sec
