A) \[{{(C{{H}_{3}})}_{2}}NH+C{{H}_{3}}COOH\]
B) \[{{(C{{H}_{3}})}_{2}}NCOOH+C{{H}_{4}}\]
C) \[2C{{H}_{3}}OH+C{{H}_{3}}CON{{H}_{2}}\]
D) \[2C{{H}_{3}}N{{H}_{2}}+C{{H}_{3}}COOH\]
Correct Answer: A
Solution :
Amides on acidic hydrolysis give acid and an amine. Hence, N-dimethylacetamide will give acetic acid and dimethyl amine on hydrolysis.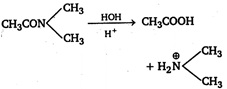
You need to login to perform this action.
You will be redirected in
3 sec
