A) trigonal bipyramidal
B) octahedral
C) tetrahedral
D) square pyramidal
Correct Answer: A
Solution :
In \[\text{Fe(CO}{{\text{)}}_{\text{5}}}\text{,}\]the Fe atom is in\[\text{ds}{{\text{p}}^{3}}\] hybridized state. Therefore, the shape of molecule is trigonal bipyramidal. The hybridisation is as \[{{\,}_{26}}Fe=1{{s}^{2}},2{{s}^{2}}2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{6}},4{{s}^{2}}4{{p}^{o}}\] In \[Fe{{(CO)}_{5}}\]the Fe atom is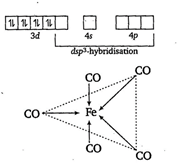
You need to login to perform this action.
You will be redirected in
3 sec
