A) \[{{\text{ }\!\![\!\!\text{ Cu(N}{{\text{H}}_{\text{3}}}{{\text{)}}_{\text{4}}}\text{ }\!\!]\!\!\text{ }}^{\text{2+}}}\] -Square planar
B) \[\text{ }\!\![\!\!\text{ Ni(CO}{{\text{)}}_{\text{4}}}\text{ }\!\!]\!\!\text{ }\] - Neutral ligand
C) \[\text{ }\!\![\!\!\text{ Fe(CN)}_{6}^{3-}\text{ }\!\!]\!\!\text{ }\] - \[\text{ }\!\![\!\!\text{ Fe(CN)}_{6}^{3-}\text{ }\!\!]\!\!\text{ s}{{\text{p}}^{3}}{{d}^{2}}\]
D) \[{{\text{ }\!\![\!\!\text{ Co(en}{{\text{)}}_{3}}\text{ }\!\!]\!\!\text{ }}^{3+}}\]- Follows EAN rule
Correct Answer: C
Solution :
(a)\[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}},Cu\] is present as \[C{{u}^{2+}}C{{u}^{2+}}=[Ar]3{{d}^{9}}4{{s}^{0}}\] \[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}=[Ar]\]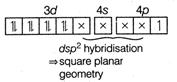 (\[N{{H}_{3}}\] being a strong field ligand shifts one electron from 3d orbital to 4p orbital.) In \[[Ni{{(CO)}_{4}}],CO\]is a neutral ligand. In\[{{[Fe{{(CN)}_{6}}]}^{3-}},\] Fe is present as\[F{{e}^{3+}}\] \[F{{e}^{3+}}=[Ar]3{{d}^{5}}4{{s}^{0}}\] \[{{[Fe(CN)6]}^{3-}}=[Ar]\]
(\[N{{H}_{3}}\] being a strong field ligand shifts one electron from 3d orbital to 4p orbital.) In \[[Ni{{(CO)}_{4}}],CO\]is a neutral ligand. In\[{{[Fe{{(CN)}_{6}}]}^{3-}},\] Fe is present as\[F{{e}^{3+}}\] \[F{{e}^{3+}}=[Ar]3{{d}^{5}}4{{s}^{0}}\] \[{{[Fe(CN)6]}^{3-}}=[Ar]\] You need to login to perform this action.
You will be redirected in
3 sec
