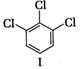
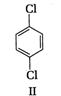
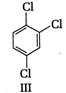
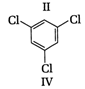
A) \[\text{I }<\text{ III }<\text{ IV }<\text{ II}\]
B) \[\text{II}=\text{IV}<\text{III}<\text{I}\]
C) \[\text{II=IIVIII}\]
D) \[\text{IIIIIIIV}\]
Correct Answer: B
Solution :
(II) and (IV) are symmetrical and thus, resultant dipole moment is zero. In (I) bond moment are towards the same direction but in (III) there is net dipole at the \[{{C}_{2}}\] position. Thus, the dipole moment of (\[I\]) is maximum.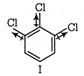
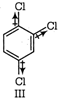 Thus. the correct order is \[\text{II = IV III I}\]
Thus. the correct order is \[\text{II = IV III I}\]
You need to login to perform this action.
You will be redirected in
3 sec
