A) cis-1, 2-dichloroethylene
B) trans-1, 2- dichloroethylene
C) 1, 1-dichloroethylene
D) none of the above
Correct Answer: B
Solution :
Key Idea: Dipole moment is a vector quantity. The molecules having symmetrical structure have zero dipole moment. [a]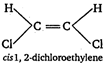 [b]
[b] 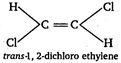 [c]
[c] 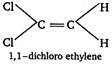 \[\because \] Out of given choices, only trans 1, 2-dichloro ethylene is symmetrical. \[\therefore \] It has zero dipole moment.
\[\because \] Out of given choices, only trans 1, 2-dichloro ethylene is symmetrical. \[\therefore \] It has zero dipole moment.
You need to login to perform this action.
You will be redirected in
3 sec
