A) \[H-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\mathop{P}}\,}}\,-OH\]
B) \[HO-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{P}}\,}}\,-OH\]
C) \[H-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\mathop{P}}\,}}\,=OH\]
D) \[H-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ OH \end{smallmatrix}}{\mathop{P}}\,}}\,-OH\]
Correct Answer: A
Solution :
Hypophosphorus acid is \[{{H}_{3}}P{{O}_{4}}\]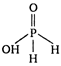 It is monobasic acid.
It is monobasic acid.
You need to login to perform this action.
You will be redirected in
3 sec
