A) Methylamine is more basic than \[N{{H}_{3}}\]
B) Amines form hydrogen bonds
C) Ethylamine has higher boiling point than propane
D) Dimethylamine is less basic than methylamine
Correct Answer: D
Solution :
As the number of electron donating groups increases, basic character increases.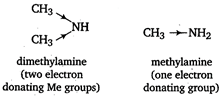 Thus, dimethylamine is more basic than methyl amine. All other statements are correct. Note Trimethyl amine however contains three electron donating groups, yet it is less basic due to steric hindrance.
Thus, dimethylamine is more basic than methyl amine. All other statements are correct. Note Trimethyl amine however contains three electron donating groups, yet it is less basic due to steric hindrance.
You need to login to perform this action.
You will be redirected in
3 sec
