A) \[{{90}^{o}}\]
B) \[{{118}^{o}}\]
C) \[{{105}^{o}}\]
D) \[{{111}^{o}}\]
Correct Answer: B
Solution :
The structure of \[Cl{{O}_{2}}\] is angular. The \[Cl\]-atom is \[s{{p}^{3}}\]-hybridised in the angular molecule with bond angle of \[{{118}^{o}}\] and \[Cl-O\]bond length of\[1.47\overset{o}{\mathop{A}}\,\]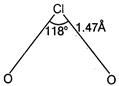
You need to login to perform this action.
You will be redirected in
3 sec
