A) nucleophilic attack, transfer of \[{{H}^{-}}\] and transfer of \[{{H}^{+}}\]
B) transfer of \[{{H}^{-}}\], transfer of \[{{H}^{+}}\] and nucleophiiic attack
C) transfer of\[{{H}^{+}}\], nucleophilic attack and transfer of\[{{H}^{-}}\]
D) electrophilic attack by \[O{{H}^{-}}\], transfer of \[{{H}^{+}}\] and transfer of \[{{H}^{-}}\]
Correct Answer: A
Solution :
The Cannizzaro reaction is as \[HCHO+HCHO\xrightarrow{KOH\,\,(con)}\underset{methyl\text{ }alcohol}{\mathop{C{{H}_{3}}OH}}\,\] \[\underset{acetic\text{ }acid}{\mathop{+HCOO{{K}^{+}}}}\,\] The mechanism of Cannizzaro reaction is as Step I Attack of nudeophile \[O{{H}^{-}}\] to the carbonyl carbon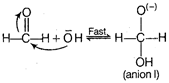 Step II The transfer of hydride ion from anion (I) to second molecule of aldehyde and finally rapid transfer of proton takes place.
Step II The transfer of hydride ion from anion (I) to second molecule of aldehyde and finally rapid transfer of proton takes place. 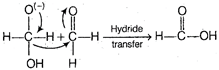 \[H-\overset{\begin{smallmatrix} O \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ H \\ alcohol \end{smallmatrix}}{\mathop{C}}\,}}\,-H\xleftarrow{Proton\text{ }exchange\text{ }fast}H-\overset{\begin{smallmatrix} {{O}^{(-)}} \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ H \\ alchol \end{smallmatrix}}{\mathop{C}}\,}}\,-{{H}^{+}}\] \[\underset{acid}{\mathop{+H-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-\overset{-}{\mathop{O}}\,\,{{K}^{+}}}}\,\]
\[H-\overset{\begin{smallmatrix} O \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ H \\ alcohol \end{smallmatrix}}{\mathop{C}}\,}}\,-H\xleftarrow{Proton\text{ }exchange\text{ }fast}H-\overset{\begin{smallmatrix} {{O}^{(-)}} \\ | \end{smallmatrix}}{\mathop{\underset{\begin{smallmatrix} | \\ H \\ alchol \end{smallmatrix}}{\mathop{C}}\,}}\,-{{H}^{+}}\] \[\underset{acid}{\mathop{+H-\overset{\begin{smallmatrix} O \\ || \end{smallmatrix}}{\mathop{C}}\,-\overset{-}{\mathop{O}}\,\,{{K}^{+}}}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
