A) \[{{[NiC{{l}_{4}}]}^{2-}}\] is square planar and \[{{[Ni{{(CN)}_{4}}]}^{2-}},\,Ni{{(CO)}_{4}}\] are tetrahedral
B) \[Ni\,{{(CO)}_{4}}\] is square planar and \[{{[Ni\,{{(CN)}_{4}}]}^{2-}},\,{{[NiC{{l}_{4}}]}^{2-}}\] are tetrahedral
C) \[{{[Ni\,{{(CN)}_{4}}]}^{2-}}\] is square planar and \[[Ni\,{{(C{{l}_{4}}]}^{2-}},Ni{{(CO)}_{4}}\] are tetrahedral
D) None of the above
Correct Answer: C
Solution :
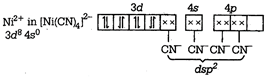

 \[{{[Ni{{(CN)}_{4}}]}^{2}}-\,ds{{p}^{2}}\to \] Square planar \[{{[NiC{{l}_{4}}]}^{2-}}-\,s{{p}^{3}}\to \] Tetrahedral \[Ni{{(CO)}_{4}}-\,s{{p}^{3}}\to \] Tetrahedral
\[{{[Ni{{(CN)}_{4}}]}^{2}}-\,ds{{p}^{2}}\to \] Square planar \[{{[NiC{{l}_{4}}]}^{2-}}-\,s{{p}^{3}}\to \] Tetrahedral \[Ni{{(CO)}_{4}}-\,s{{p}^{3}}\to \] Tetrahedral
You need to login to perform this action.
You will be redirected in
3 sec
