Directions : In the following question more than one of the answers given may be correct. Select the correct answer and mark it according to the code:
The compounds show the geometrical isomerism are: (1)A) 1, 2 and 3 are correct
B) 1 and 2 are correct
C) 2 and 4 are correct
D) 1 and 3 are correct
Correct Answer: C
Solution :
A compound containing a double bond can show geometrical isomerism only when identical groups are not present on the double bonded carbon.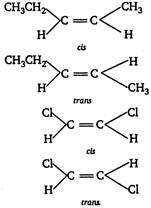
You need to login to perform this action.
You will be redirected in
3 sec
