Directions : In the following question more than one of the answers given may be correct. Select the correct answer and mark it according to the code:
The wavelengths and frequencies of photons in transitions 1, 2 and 3 for hydrogen like atom are\[{{\lambda }_{1}},{{\lambda }_{2}},{{\lambda }_{3}};{{v}_{1}},{{v}_{2}}\]and\[{{v}_{3}}\]respectively. Then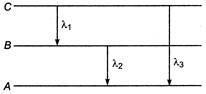 (1) \[{{v}_{3}}={{v}_{1}}+{{v}_{2}}\] (2) \[{{\lambda }_{3}}={{\lambda }_{1}}+{{\lambda }_{2}}\] (3) \[{{\lambda }_{3}}=\frac{{{\lambda }_{1}}{{\lambda }_{2}}}{{{\lambda }_{1}}+{{\lambda }_{2}}}\] (4) \[{{v}_{3}}=\frac{{{v}_{1}}{{v}_{2}}}{{{v}_{1}}+{{v}_{2}}}\]
(1) \[{{v}_{3}}={{v}_{1}}+{{v}_{2}}\] (2) \[{{\lambda }_{3}}={{\lambda }_{1}}+{{\lambda }_{2}}\] (3) \[{{\lambda }_{3}}=\frac{{{\lambda }_{1}}{{\lambda }_{2}}}{{{\lambda }_{1}}+{{\lambda }_{2}}}\] (4) \[{{v}_{3}}=\frac{{{v}_{1}}{{v}_{2}}}{{{v}_{1}}+{{v}_{2}}}\]
A) 1, 2 and 3 are correct
B) 1 and 2 are correct
C) 2 and 4 are correct
D) 1 and 3 are correct
Correct Answer: D
Solution :
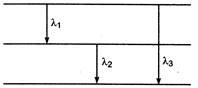 From figure \[{{E}_{3}}={{E}_{1}}+{{E}_{2}}\] \[h{{v}_{3}}=h{{v}_{1}}+h{{v}_{2}}\] Again, \[{{v}_{3}}={{v}_{1}}+{{v}_{2}}\] \[\frac{hc}{{{\lambda }_{3}}}=\frac{hc}{{{\lambda }_{1}}}+\frac{hc}{{{\lambda }_{2}}}\] \[\frac{1}{{{\lambda }_{3}}}=\frac{1}{{{\lambda }_{1}}}+\frac{1}{{{\lambda }_{2}}}\] \[\frac{1}{{{\lambda }_{3}}}=\frac{{{\lambda }_{2}}+{{\lambda }_{1}}}{{{\lambda }_{1}}{{\lambda }_{2}}}\] \[\lambda {{ & }_{3}}=\frac{{{\lambda }_{1}}{{\lambda }_{2}}}{{{\lambda }_{1}}+{{\lambda }_{2}}}\] Hence, option is correct.
From figure \[{{E}_{3}}={{E}_{1}}+{{E}_{2}}\] \[h{{v}_{3}}=h{{v}_{1}}+h{{v}_{2}}\] Again, \[{{v}_{3}}={{v}_{1}}+{{v}_{2}}\] \[\frac{hc}{{{\lambda }_{3}}}=\frac{hc}{{{\lambda }_{1}}}+\frac{hc}{{{\lambda }_{2}}}\] \[\frac{1}{{{\lambda }_{3}}}=\frac{1}{{{\lambda }_{1}}}+\frac{1}{{{\lambda }_{2}}}\] \[\frac{1}{{{\lambda }_{3}}}=\frac{{{\lambda }_{2}}+{{\lambda }_{1}}}{{{\lambda }_{1}}{{\lambda }_{2}}}\] \[\lambda {{ & }_{3}}=\frac{{{\lambda }_{1}}{{\lambda }_{2}}}{{{\lambda }_{1}}+{{\lambda }_{2}}}\] Hence, option is correct.
You need to login to perform this action.
You will be redirected in
3 sec
