A) crystallization
B) fractional crystallization
C) distillation
D) steam distillation
Correct Answer: D
Solution :
o-nitrophenol has intra molecular hydrogen bonding, while p-nitrophenol has intermolecular hydrogen bonding (comparitively stronger). Due to this reason, the boiling point of o-nitrophenol is found quite less than that of p-nitrophenol. Hence, o-nitrophenol is steam volatile and can be separated from p-nitrophenol by steam distillation.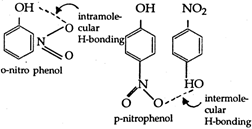
You need to login to perform this action.
You will be redirected in
3 sec
