A) 0.922 per minute
B) 0.691 per minute
C) 0.461 per minute
D) 0.230 per minute
Correct Answer: C
Solution :
According to law of radioactivity \[\frac{N}{{{N}_{0}}}={{e}^{-\lambda t}}\] ??.(i) Taking logarithm on both sides of Eq. (i), we have \[{{\log }_{e}}\left( \frac{N}{{{N}_{0}}} \right)={{\log }_{e}}({{e}^{-\lambda t}})\] \[=-\lambda t\,{{\log }_{e}}\,e=-\lambda t\] As we know that \[{{\log }_{e}}\,x=2.3026\,\,{{\log }_{10}}x\] Making substitution, we get \[\lambda =\frac{2.3026\,\,{{\log }_{10}}\,\,\left( \frac{9750}{975} \right)}{5}\] \[=\frac{2.3026}{e}\,{{\log }_{10}}10\] \[=\frac{2.3026}{5}\,{{\min }^{-1}}=0.461\,{{\min }^{-1}}\] NOTE: 1. The graph between number of nuclei decayed \[({{N}_{0}}-N)\] with time (t) is as shown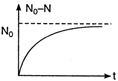 The graph between probability of a nucleus for survival \[\left( \frac{N}{{{N}_{0}}} \right)\] with time (f) is as shown below.
The graph between probability of a nucleus for survival \[\left( \frac{N}{{{N}_{0}}} \right)\] with time (f) is as shown below. 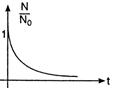
You need to login to perform this action.
You will be redirected in
3 sec
