A) \[M{{g}^{2+}}\]
B) \[C{{a}^{2+}}\]
C) \[C{{u}^{+}}\]
D) \[C{{u}^{2+}}\]
Correct Answer: D
Solution :
Borax bead test is given by only those transition metal ions or salts which contain unpaired electrons. Among the given ions, only \[C{{u}^{+}}\] and \[C{{u}^{2+}}\] are transition metal ions. Out of these only \[C{{u}^{2+}}\] contains unpaired electron, hence it gives borax bead test.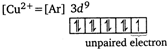
You need to login to perform this action.
You will be redirected in
3 sec
