A) Ethanol is more acidic than phenol
B) Phenol is more acidic than ethanol
C) Phenol is more acidic than benzoic acid
D) Acidity of phenol and benzoic acid is about equal
Correct Answer: B
Solution :
Phenol is more acidic than ethanol due to resonance stabilisation of phenoxide ion.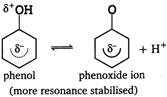 \[\underset{(no\,resonance)}{\mathop{R-OHR-{{O}^{-}}+{{H}^{+}}}}\,\]
\[\underset{(no\,resonance)}{\mathop{R-OHR-{{O}^{-}}+{{H}^{+}}}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
