A) bromine in benzene
B) bromine in water
C) \[KBr\] solution
D) \[B{{r}_{2}}\] in \[CC{{l}_{4}}\] at \[{{0}^{o}}C\]
Correct Answer: B
Solution :
X is bromine in water.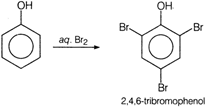 With aqueous , bromination occurs at both the o- and one p-positions to give 2,4,6-tribromophenol while with \[B{{r}_{2}}\] in \[CC{{l}_{4}}\], bromination occurs mainly at the less hindered p-position to give p-bromophenol as the major product.
With aqueous , bromination occurs at both the o- and one p-positions to give 2,4,6-tribromophenol while with \[B{{r}_{2}}\] in \[CC{{l}_{4}}\], bromination occurs mainly at the less hindered p-position to give p-bromophenol as the major product.
You need to login to perform this action.
You will be redirected in
3 sec
