A) linear
B) planar
C) pyramidal
D) T-shaped
Correct Answer: B
Solution :
In \[{{X}_{e}}{{O}_{3}},\], total electron pairs \[=4(3bp+1/p)\] i.e., \[s{{p}^{3}}\] hybridisation \[\Rightarrow \] pyramidal shape.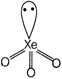
You need to login to perform this action.
You will be redirected in
3 sec
