A) catalytic hydrogenation of alkenes
B) Wurtz reaction
C) hydrolysis of alkyl magnesium bromide
D) Kolbes electrolytic method
E) dehydrohalogenation of an alkyl halide
Correct Answer: E
Solution :
Dehydrohalogenation of an alkyl halide does not give an alkane because it gives alkenes as,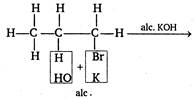 \[H-\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,-C=\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}\,-H+KBr+{{H}_{2}}O\]
\[H-\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}}\,-C=\overset{\begin{smallmatrix} H \\ | \end{smallmatrix}}{\mathop{C}}\,-H+KBr+{{H}_{2}}O\]
You need to login to perform this action.
You will be redirected in
3 sec
