| 1. phenol |
| 2. benzyl alcohol |
| 3. benzyl iodide |
| 4. iodobenzene |
A) 1 and 3 only
B) 3 and 4 only
C) 1 and 4 only
D) 2 and 4 only
E) 2 and 3 only
Correct Answer: A
Solution :
Benzyl phenyl ether is an unsymmetrical ether, so halide ion of HI is attached to the simple alkyl group and the reaction takes place by following mechanism: (i) Protonation of ether \[\underset{benzyl\text{ }phenyl\text{ }ether}{\mathop{{{C}_{6}}{{H}_{5}}C{{H}_{2}}O{{C}_{6}}{{H}_{5}}}}\,+HI\to {{C}_{6}}{{H}_{5}}C{{H}_{2}}-\underset{\begin{smallmatrix} | \\ H \end{smallmatrix}}{\overset{\begin{smallmatrix} \oplus \\ \bullet \,\bullet \end{smallmatrix}}{\mathop{O}}}\,-{{C}_{6}}{{H}_{5}}+{{I}^{-}}\] (ii) Nucleophilic attack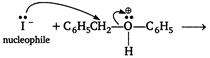 \[\underset{benzyl\text{ }iodide}{\mathop{{{C}_{6}}{{H}_{5}}C{{H}_{2}}I}}\,+\underset{phenol}{\mathop{{{C}_{6}}{{H}_{5}}OH}}\,\]
\[\underset{benzyl\text{ }iodide}{\mathop{{{C}_{6}}{{H}_{5}}C{{H}_{2}}I}}\,+\underset{phenol}{\mathop{{{C}_{6}}{{H}_{5}}OH}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
