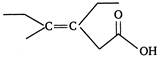
A) Z-3-ethyl-4-methyl hex-3-en-1-oic acid
B) Z-3-ethyl-4-methyl hexanoic acid
C) Z-3, 4-diethylpent-3-en-1-oic acid
D) Z-3-ethyl-4-methylhex-4-en-1-oic acid
E) E-3-ethyl-4-methylhex-3-en-1-oic acid
Correct Answer: E
Solution :
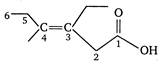 E-3-ethyl-4-methylhex-3-en-l-oic acid. [The configuration of this compound is E because bulkier groups are present at opposite sides of the double bond.]
E-3-ethyl-4-methylhex-3-en-l-oic acid. [The configuration of this compound is E because bulkier groups are present at opposite sides of the double bond.]
You need to login to perform this action.
You will be redirected in
3 sec
