A) 1-butanol and 2-butanol
B) cis-2-butene and trans-2-butene
C) dimethyl ether and ethanol
D) acetone and propanal
E) ethanol and ethanol
Correct Answer: B
Solution :
But-2-ene shows stereoisomerism and can exist in the following two forms: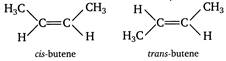 Both these isomers have the same structural formulae but differ in the relative spatial arrangement of hydrogen atoms and methyl groups around the double bond.
Both these isomers have the same structural formulae but differ in the relative spatial arrangement of hydrogen atoms and methyl groups around the double bond.
You need to login to perform this action.
You will be redirected in
3 sec
