A) geometric, optical, position and functional isomerism
B) geometric, optical and functional isomerism
C) position and functional isomerism only
D) geometric and optical isomerism only
E) geometric isomerism only
Correct Answer: A
Solution :
(i) Geometrical isomers of compound \[CHCl=CHCHOHCOOH\]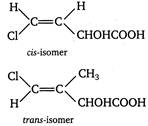 (ii) It also shows optical isomerism as
(ii) It also shows optical isomerism as 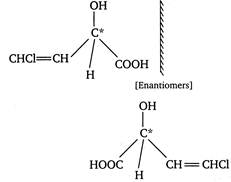 (iii) It can also show position isomerism. For example two positional isomers are given below: \[\underset{4\text{ }-chloro-2-hydroxy\text{ }-3-butene\text{ }-1\text{ }-oic\text{ }acid}{\mathop{\underset{4}{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{CH}}}\,=\underset{3}{\mathop{C}}\,H-\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{\underset{2}{\mathop{C}}\,H}}\,-\underset{1}{\mathop{C}}\,OOH}}\,\] \[\underset{\begin{smallmatrix} 4-chloro-3-hydroxy-3-butene \\ \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,-1-oic\text{ }acid \end{smallmatrix}}{\mathop{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{CH}}\,=\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{2}}-COOH}}\,\] (iv) It can also show functional isomerism as functional isomer of an alcohol is ether. \[\underset{alcohol}{\mathop{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{CH}}\,=CH\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{CH}}\,COOH}}\,\] \[\underset{ether}{\mathop{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{CH}}\,=CHO\text{ }C{{H}_{2}}\text{ }COOH}}\,\]
(iii) It can also show position isomerism. For example two positional isomers are given below: \[\underset{4\text{ }-chloro-2-hydroxy\text{ }-3-butene\text{ }-1\text{ }-oic\text{ }acid}{\mathop{\underset{4}{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{CH}}}\,=\underset{3}{\mathop{C}}\,H-\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{\underset{2}{\mathop{C}}\,H}}\,-\underset{1}{\mathop{C}}\,OOH}}\,\] \[\underset{\begin{smallmatrix} 4-chloro-3-hydroxy-3-butene \\ \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,-1-oic\text{ }acid \end{smallmatrix}}{\mathop{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{CH}}\,=\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{C}}\,-C{{H}_{2}}-COOH}}\,\] (iv) It can also show functional isomerism as functional isomer of an alcohol is ether. \[\underset{alcohol}{\mathop{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{CH}}\,=CH\overset{\begin{smallmatrix} OH \\ | \end{smallmatrix}}{\mathop{CH}}\,COOH}}\,\] \[\underset{ether}{\mathop{\overset{\begin{smallmatrix} Cl \\ | \end{smallmatrix}}{\mathop{CH}}\,=CHO\text{ }C{{H}_{2}}\text{ }COOH}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
