A) \[Mg+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}MgO\]
B) \[2Ag+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}A{{g}_{2}}O\]
C) \[C+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}CO\]
D) \[CO+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}C{{O}_{2}}\]
Correct Answer: A
Solution :
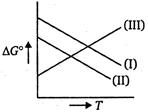 Reaction (i) \[M(s)+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}MO(s)\] Reaction (ii) \[CO(g)+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}C{{O}_{2}}(g)\] Reaction (in) \[C(s)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}C{{O}_{2}}(g)\] \[\Delta {{G}^{o}}vs\],T plot in the Ellinghams diagram slopes downwards for the reaction (I) and reaction (II). For reaction (III) \[\Delta {{G}^{o}}vs\,T\] m the diagram slopes upwards.
Reaction (i) \[M(s)+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}MO(s)\] Reaction (ii) \[CO(g)+\frac{1}{2}{{O}_{2}}\xrightarrow{{}}C{{O}_{2}}(g)\] Reaction (in) \[C(s)+\frac{1}{2}{{O}_{2}}(g)\xrightarrow{{}}C{{O}_{2}}(g)\] \[\Delta {{G}^{o}}vs\],T plot in the Ellinghams diagram slopes downwards for the reaction (I) and reaction (II). For reaction (III) \[\Delta {{G}^{o}}vs\,T\] m the diagram slopes upwards.
You need to login to perform this action.
You will be redirected in
3 sec
