A) methanol and bromoethane
B) ethyl hydrogen sulphate and alcoholic KOH
C) ethyl hydrogen sulphate and aqueous KOH
D) ethanol and alcoholic KOH
Correct Answer: D
Solution :
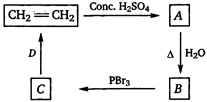
\[C{{H}_{2}}=C{{H}_{2}}\xrightarrow{Cone.\text{ }{{H}_{2}}S{{O}_{4}}}\underset{(addition\text{ }reaction)}{\mathop{C{{H}_{3}}-\underset{(A)}{\mathop{C{{H}_{2}}}}\,-HS{{O}_{4}}}}\,\] \[C{{H}_{3}}-C{{H}_{2}}-HS{{O}_{4}}\xrightarrow{\Delta ,\,\,{{H}_{2}}O}\underset{(hydrolysis)}{\mathop{C{{H}_{3}}-\underset{(B)}{\mathop{C{{H}_{2}}}}\,-OH}}\,\]\[C{{H}_{3}}-C{{H}_{2}}-OH\xrightarrow{PB{{r}_{3}}}\underset{(bromination)}{\mathop{C{{H}_{3}}-\underset{(C)}{\mathop{C{{H}_{2}}}}\,-Br}}\,\] \[C{{H}_{3}}-C{{H}_{2}}-Br\xrightarrow[(\Delta )]{AlC.\,KOH\,(\Delta )}C{{H}_{2}}=C{{H}_{2}}\] (dehydrohalogenation) Hence, B and D in the above given reactions are \[C{{H}_{3}}C{{H}_{2}}OH\](ethanol) and ale. \[KOH\].You need to login to perform this action.
You will be redirected in
3 sec
