A) \[{{[Co{{(N{{H}_{3}})}_{6}}]}^{2+}}\] is paramagnetic
B) \[{{[MnB{{r}_{4}}]}^{2-}}\] is tetrahedral
C) \[{{[CoB{{r}_{2}}{{(en)}_{2}}]}^{-}}\] exhibits linkage isomerism
D) \[{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}\] is an inner orbital complex.
Correct Answer: A
Solution :
: [a] \[{{[Co{{(N{{H}_{3}})}_{6}}]}^{2+}}\] \[_{27}C{{o}^{2+}}:[Ar]\,\,3{{d}^{7}}\,4{{s}^{0}}\]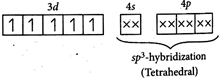 [c] \[{{[CoB{{r}_{2}}{{(en)}_{2}}]}^{-}}\]exhibit geometrical isomerism.
[c] \[{{[CoB{{r}_{2}}{{(en)}_{2}}]}^{-}}\]exhibit geometrical isomerism. 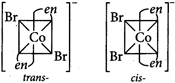 [d] \[{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}\] \[_{28}N{{i}^{2+}}:\,\,[Ar]\,\,3{{d}^{8}}\]
[d] \[{{[Ni{{(N{{H}_{3}})}_{6}}]}^{2+}}\] \[_{28}N{{i}^{2+}}:\,\,[Ar]\,\,3{{d}^{8}}\] 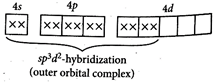 Both statements [a] and [b] are correct.
Both statements [a] and [b] are correct.
You need to login to perform this action.
You will be redirected in
3 sec
