A) Triangular
B) Square planar
C) Tetrahedral
D) Hexagonal
Correct Answer: C
Solution :
In methane the central atom, carbon is in a state of \[s{{p}^{3}}\]hybridization. Four \[s{{p}^{3}}\] hybrid orbitals around the central atom are directed tetrahedrally in a bid to be at maximum possible distance from each other. These four tetrahedrally inclined orbitals overlap with s-orbitals of four H-atom. The four \[C-H\] bonds are mutually inclined at an angle of\[{{109}^{o}}28'\]. Tetrahedral shape of methane molecule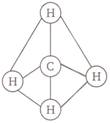
You need to login to perform this action.
You will be redirected in
3 sec
