A) \[n=3,l=1,m=0,s=-1/2\]
B) \[n=4,l=2,m=0,s=+1/2\]
C) \[n=3,l=1,m=+1,s=+1/2\]
D) \[n=2,l=0,m=0,s=+1/2\]
Correct Answer: C
Solution :
Electronic configuration of chlorine is \[_{17}Cl=1{{s}^{2}},2{{p}^{6}},3{{s}^{2}}3{{p}^{5}}\]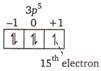 \[n=3\] \[l=1\] \[m=+1,\] and \[s=+1/2\]
\[n=3\] \[l=1\] \[m=+1,\] and \[s=+1/2\]
You need to login to perform this action.
You will be redirected in
3 sec
