A) \[2\]
B) \[1\]
C) \[0\]
D) \[3\]
Correct Answer: C
Solution :
The electronic configuration of \[Pt=[Xe]4{{f}^{14}},5{{d}^{9}},6{{s}^{1}}\] \[\therefore \] \[P{{t}^{2+}}=[Xe]4{{f}^{14}},5{{d}^{8}},6{{s}^{0}}\] \[{{[Pt{{(CN)}_{4}}]}^{2-}}=[Xe]4{{f}^{14}}\]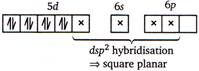 \[\therefore \] No unpaired electron is present in \[{{[Pt{{(CN)}_{4}}]}^{2-}}\] ion.
\[\therefore \] No unpaired electron is present in \[{{[Pt{{(CN)}_{4}}]}^{2-}}\] ion.
You need to login to perform this action.
You will be redirected in
3 sec
