(I) 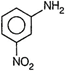 | (II) |
| (III) | (IV) |
A) \[I<II<III<IV\]
B) \[IV<II<III<II\]
C) \[I<II<IV<III\]
D) \[IV<III<I<II\]
Correct Answer: A
Solution :
Presence of electron donating groups like\[-OC{{H}_{3}}\] and \[-C{{H}_{3}}\]increase the basicity of amines whereas, presence of electron withdrawing. group like \[-N{{O}_{2}}\] decrease the basicity of amines, Hence, basicity increases in the order I < II < III < IV.You need to login to perform this action.
You will be redirected in
3 sec
